The science of pharmacogenomics
Where genes and medications meet
Pharmacogenomics (PGx) is a branch of precision medicine dedicated to understanding how genetic variants contribute to different responses to the same drug in different patients. It is gaining use across the care continuum and in specialties ranging from primary care/family medicine to psychiatry to oncology.
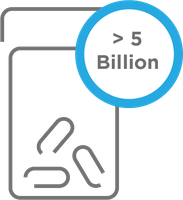
One size doesn’t fit all
Over 5 billion prescriptions are filled each year.1 However, not all drugs are effective for all people. In fact, response rates for many drugs are only 50-75%.2 One of the factors that can help explain this variability is an individual patient's DNA, which may affect drug metabolism (pharmacokinetics), mechanism of action (pharmacodynamics), and drug toxicity.
How DNA affects medication response
Pharmacokinetics
Certain genes influence drug metabolism, which may significantly impact how much of a medication your body may be exposed to. Patients who process medications slower or faster than normal may have increased or decreased exposure to a medication, which may lead to a dramatically different response to the same medication and dosage.
Pharmacodynamics
Certain genes can influence the function of proteins responsible for the drug’s mechanism of action and in turn impact medication response. Drug targets, which include transporters, receptors, and other non-metabolizing enzymes, are considered pharmacodynamic genes.
Additional PGx impacts
Certain genes carry variants that have been linked to risk of adverse reactions. Most notably certain alleles of the human leukocyte antigen (HLA) genes have been linked to severe drug hypersensitivity reactions.3

Evidence for PGx in patient care
PGx testing provides information that healthcare teams may use to assess the impact of an individual’s DNA on medication related outcomes, and take into consideration in the course of care. With adverse drug reactions estimated to be the 4th leading cause of death in the U.S.4 and prescription spending on an invoice basis at $479 billion in 2018,1 this information can offer significant value to patient care.
Supporting facts
> 90% of patients possess clinically actionable genetic variants.5,6
The FDA has an extensive list of pharmacogenomic biomarkers for drugs on the market today.3
Adverse drug reactions result in 1.3 million emergency department visits annually, approximately 350,000 of which require hospital admission.7
Clinical data curation
At OneOme, our clinical development process is extensive and incorporates both scientific and clinical data. We are driven to help healthcare providers optimize medication therapy by providing a credible, robust, and comprehensive pharmacogenomic solution.
Our data and clinical annotation curation is performed by a team of clinical pharmacists and scientists. The relationships between medications and pharmacogenes annotated in our database are supported by scientific evidence that meets OneOme’s rigorous inclusion criteria. For more information, view our Indications for use.
Provider considerations are curated by the OneOme clinical development team by extensive literature evaluation, including published primary literature, US Food and Drug Administration (FDA) label excerpts, and professional guidelines published by Clinical Pharmacogenetics Implementation Consortium (CPIC) and Dutch Pharmacogenomics Working Group (DPWG).
References
- IQVIA Institute for Human Data Science. Medicine Use and Spending in the U.S. https://www.iqvia.com/insights/the-iqvia-institute/reports/medicine-use-and-spending-in-the-us-a-review-of-2018-and-outlook-to-2023 Published May 2019. Accessed June 10, 2020.
- Spear BB, Heath-Chiozzi M, Hu J. Clinical application of pharmacogenetics. Trends Mol. Med. 2001; 7, 201–204.
- Table of Pharmacogenomic Biomarkers in Drug Labeling. U.S. Food and Drug Administration website.: https://www.fda.gov/drugs/science-and-research-drugs/table-pharmacogenomic-biomarkers-drug-labelinghttps://www.fda.gov/Drugs/ScienceResearch/ucm572698.htm Accessed June 10, 2020.
- Preventable Adverse Drug Reactions: A Focus on Drug Interactions. U.S. Food and Drug Administration website. www.fda.gov/Drugs/DevelopmentApprovalProcess/DevelopmentResources/DrugInteractionsLabeling/ucm110632.htm. Accessed June 10, 2020.
- Van Driest SL, Shi Y, Bowton EA, et al. Clinically actionable genotypes among 10,000 patients with preemptive pharmacogenomic testing. Clin Pharmacol Ther. 2014; 95(4):423‐431.
- Yuan, J. et. al. Preemptive Pharmacogenomic Testing for Precision Medicine. J Mol Diag. 2016; 18(3): 438-445.
- Adverse Drug Events in Adults. Centers for Disease Control and Prevention website. Last reviewed October 11, 2017. Accessed June 10, 2020.